Figures
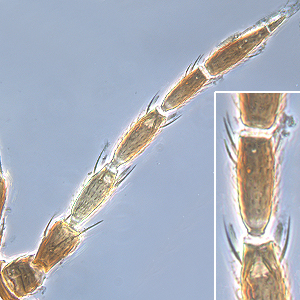
Fig.�1: Antenna (inset: III. and�IV. antennal segment)
Fig. 2: Head dorsal with ocellar triangle
Fig. 3: Pronotum
Fig. 4: Meso- and metanotum
Fig. 5: Forewing
Fig. 6: Sternites VI and VII
Fig. 7: Tergites V and VI
Fig. 8:�Tergites VIII and IX
ITS-RFLP gel patterns (1&8 ladder, 2 PCR-product, 3 RSAI, 4 HaeIII, 5 MspI, 6 HinfI, 7 AluI)
Fig. 9: Primer pair O1/18J
Fig. 10: Primer pair 18SMP/28SMP
Taxonomic Information
Species:
Thrips hawaiiensis (Morgan, 1913)
Synonyms:
Taeniothrips florinatus Priesner, 1938
Taeniothrips rhodomyrti Priesner, 1938
Thrips imitator Priesner, 1934
Physothrips darci Girault, 1930
Physothrips lacteicolor Girault, 1928
Physothrips marii Girault, 1928
Taeniothrips eriobotryae Moulton, 1928
Physothrips emersoni Girault, 1927
Thrips io Girault, 1927
Thrips partirufus Girault, 1927
Thrips pallipes Bagnall, 1926
Thrips versicolor Bagnall, 1926
Bregmatothrips theifloris Karny, 1921
Physothrips pallipes Bagnall, 1916
Physothrips albipes Bagnall, 1916
Thrips albipes Bagnall, 1914
Euthrips hawaiiensis Morgan, 1913
Thrips nigriflava Schmutz, 1913
Thrips sulphurea Schmutz, 1913
Common name:
Banana flower thrips
Present taxonomic position:
Family: Thripidae Stephens, 1829
Subfamily: Thripinae (Stephens) Karny, 1921
Genus: Thrips Linneaeus, 1758
Species Recognition
General information about the genus Thrips:There are about 280 species currently recognized in the genus Thrips making this genus one of the largest groups within the Thysanoptera. They are separated from other genera in having the following characters, antenna comprising 7 or�8 segments with segments III and IV containing forked sense cones, the head has two pairs of ocellar setae (II and III), pair I is missing, the pronotum with four long setae on the posterior margin, forewing 1st vein usually has a row of setae interrupted by gaps, on lateral sides of abdominal tergites V to VIII there are paired ctenidia, abdominal tergite VIII with ctenidia posterior to�the spiracles.
Typical character states of Thrips hawaiiensis:
Body color
Mainly brown�or�bicolored
Antennae
Number of antennal segments: 7-8
Segment IV - forked sensorium:�scarcely extending beyond base of segment V
Segments II and III shape:�more or less symmetric
Segments III & IV sensoria:�emergent and forked
Base of sensorium on antennal segment VI:�no more than 2 times as wide as base of nearest seta
Terminal antennal segments:�rarely elongate
Head:
Distance between bases of ocellar setae III:�greater than width of first ocellus
Head shape between compound eyes:�not prolonged
Ocellar setae III on head:�arising on anterior margin of, or in front of, ocellar triangle
Postocular setae I:�absent
Surface of head, pronotum and fore legs:�without strong reticulate sculpture
Ocellar setae I in front of anterior ocellus:�absent
Prothorax
Number of pairs of elongate pronotal setae: 0-3
Number of pairs of elongate posteroangular pronotal setae: 2
Pronotum shape:�rectangular
Number of pairs of pronotum posteromarginal minor setae: 3-4
Number of pairs of pronotum anteromarginal minor setae: 5-6
Mesothorax
Mesothoracic endofurca:�with median spinula
Metathorax
Metanotal median area sculptured lines:�transverse at anterior, but longitudinal and parallel on posterior half
Metanotal median setae length:�longer than lateral metanotal setae
Metanotal median setae position:�arising at anterior margin
Metanotum:�without campaniform sensilla
Metanotum major sclerite:�with two major sclerites, metascutum and metascutellum
Metanotum median area:�with no equiangular reticulation
Metanotum sculpture:�without dominant sculptured triangle medially
Metathoracic endofurca:�transverse, sometimes with simple median spinula
Wings
Wings:�present and more than half as long as abdomen
First vein of forewing:�distinct from costal vein
Forewing anterior margin:�with setae and cilia but cilia longer than setae
Forewing clavus:�terminal veinal seta longer than subterminal seta
Forewing color:�uniformly dark or shaded, but with base (or sub-base) pale�or�uniformly light brown
Forewing costal fringe of cilia:�arising at anterior margin of wing
Forewing costal setae at middle of wing:�shorter than median width of wing
Forewing first vein setal row:�incomplete, with setae not closely and uniformly spaced
Forewing posterior margin cilia:�undulated near apex
Forewing second vein setal row:�complete, with setae closely and uniformly spaced
Forewing surface:�not reticulate
Forewings:�with veins, setae and microtrichia
Legs
Fore tarsus inner apex:�without tooth
Fore tibial apex:�not extending around fore tarsus -�with small curved claw ventrolaterally
Mid and hind tarsi:�with two segments
Abdomen:
Pleurotergal discal setae:�absent
Abdominal pleurotergites:�not covered in microtrichia
Abdominal segment X:�never tubular, longitudinally incomplete ventrally in both sexes
Abdominal sternite II:�with 1 or 2 discal setae in addition to marginal setae or�with marginal setae but no discal setae
Abdominal sternite III of female:�without glandular areas
Abdominal sternite VII:�with discal setae present on median area
Abdominal sternite VII median marginal setae:�arising in front of margin
Abdominal sternites IV , V and VI:�with discal setae present medially as well as marginal setae
Number of lateral marginal setae on abdominal tergite II: 4
Abdominal tergites:�without curved wing-retaining setae
Abdominal tergites IV & V median setal pair:�much shorter than distance between their bases
Abdominal tergites V-VII:�with pair of ctenidia laterally
Number of discal setae on sternite V: 10-13
Setae on abdominal tergite X:�slender
Surface of lateral thirds of abdominal tergites:�without regular rows of fine microtrichia
Ctenidia on tergite VIII:�posteromesad to spiracle
Tergite VIII posteromarginal comb of microtrichia:�present, complete medially
Tergite VIII posteromarginal microtrichia:�short and irregular in length
Biology
Life history:
As with other thrips species the life cycle from egg to adult is dependent on�temperature.�The full cycle can take about 15 days (Lewis, 1973) to over a month and adults may live for more than one month producing several generations in one year depending on seasonal weather. With greenhouse temperatures the developmental time from egg to adult can decrease to about one week.
Host plants:
Polyphagous, flower feeding
Vector capacity:
None identified
Current known distribution:
Asia, Australia, New Zealand, North America
Additional notes:
Thrips hawaiiensis is difficult to separate from T. florum however Nakahara (1994) provides an adequate comparison of the two species based on setal length and presence. In addition, T. hawaiiensis has sculpture on the mesonotum by the anteromedial pores whereas T. florum is not sculptured. Thrips hawaiiensis has been found in flowers of a wide range of host plants.
Bibliography
Abe, M & Ikegami, T (2005): Susceptibility of five species of thrips to different strains of the entomopathogenic fungus, Beauveria bassiana. Applied Entomology and Zoology, 40, 667-674.
Bailey, SF (1957): The thrips of
California Part I: Suborder Terebrantia. Bulletin of the California Insect
Survey 4, no. 5: 143-220.
Bhatti, JS (1999): New characters for identification of the pest species Thrips hawaiiensis and florum (Terebrantia: Thripidae). Thrips 1: 31-53. [Scientia Publishing, New Delhi].
Childers, CC & Nakahara, S (2006): Thysanoptera (thrips) within citrus orchards in Florida: Species distribution, relative and seasonal abundance within trees, and species on vines and ground cover plants. Journal of Insect Science, 6.
Imai, T, Maekawa, M & Murai, T (2001): Attractiveness of methyl anthranilate and its related compounds to the flower thrips, Thrips hawaiiensis (Morgan), T. coloratus Schmutz, T. flavus Schrank and Megalurothrips distalis (Karny) (Thysanoptera : Thripidae). Applied Entomology and Zoology, 36, 475-478.
Inoue, T, Sakurai, T, Murai, T & Maeda, T (2004): Specificity of accumulation and transmission of tomato spotted wilt virus (TSWV) in two genera, Frankliniella and Thrips (Thysanoptera : Thripidae). Bulletin of Entomological Research, 94, 501-507.
Lewis, T (1973): Thrips their biology, ecology and economic
importance. Academic Press Inc., London Ltd. 349 pp.
Moritz G, Morris DC, Mound LA (2001): ThripsID -
Pest thrips of the world. ACIAR
and CSIRO Publishing Collingwood, Victoria, Australia, CDROM ISBN 1
86320 296 X.
Moritz G, Mound LA, Morris DC, Goldarazena A (2004): Pest
thrips of the world - an identification and information system using molecular
and microscopial methods. CBIT, University of Queensland,CDROM ISBN 1-86499-781-8.
Mound,
LA & Kibby, G (1998): Thysanoptera: An identification guide, (2nd
edition). CAB International, Wallingford and New York, 70pp.
Mound, LA & Marullo, R (1996): The
thrips of Central and South America: An Introduction (Insecta: Thysanoptera).
Associated Publishers, Gainesville.
Murai, T, Imai, T & Maekawa, M (2000): Methyl anthranilate as an attractant for two thrips species and the thrips parasitoid Ceranisus menes. Journal of Chemical Ecology, 26, 2557-2565.
Murai, T (2001): Development and reproductive capacity of Thrips hawaiiensis (Thysanoptera : Thripidae) and its potential as a major pest. Bulletin of Entomological Research, 91, 193-198.
Nakahara, S (1985): Review of Thrips hawaiiensis and revalidation of T. florum (Thysanoptera: Thrpidae). Proceedings of the Entomological Society of Washington, 87:864-870.
Nakahara, S (1994): The Genus Thrips Linnaeus (Thysanoptera: Thripidae) of the New World. USDA Agricultural Research Service Technical bulletin No. 1822.
Palmer, JM & Wetton, MN (1987): A morphometric analysis of the Thrips hawaiiensis (Morgan) species-group (Thysanoptera, Thripidae). Bulletin of Entomological Research, 77, 397-406.
Raizada, U & Saxena, P (1989): Experimental studies in cross pollination of radish (Raphanus sativus L.) by Thrips flavus Schrank and Thrips hawaiiensis (Morgan) (Thysanoptera: Insecta). Ecology (Journal of Pure and Applied Entomology), 2, 111-119.
Stannard, LJ (1968): The thrips, or Thysanoptera, of Illinois. Illinois Natural History Survey Bulletin 29: 215-552.
Links:
Mound, LA (2005): Thysanoptera (Thrips) of the World
- A Checklist. http://www.ento.csiro.au/thysanoptera/worldthrips.html